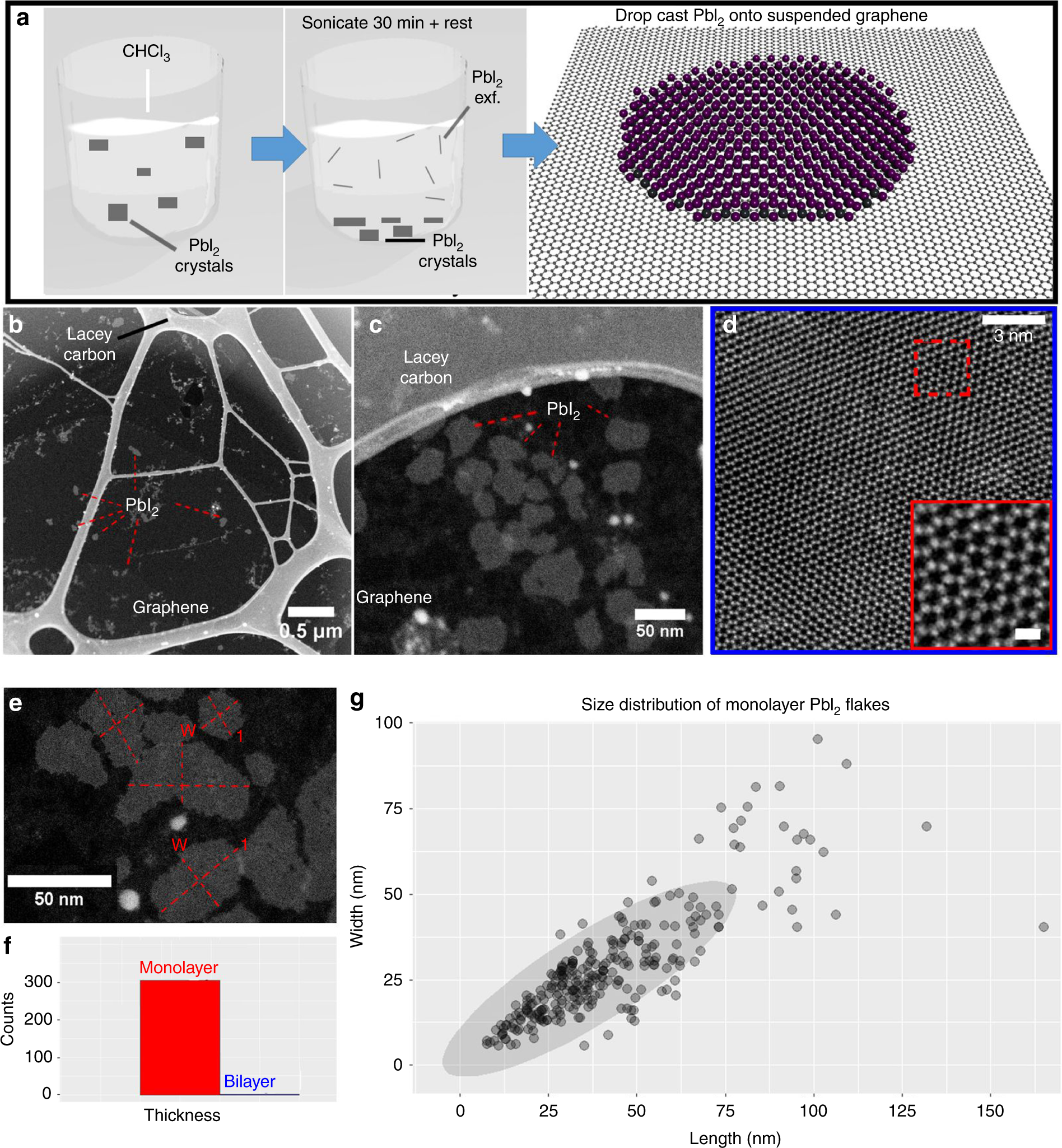
In the Museum's collection are models that demonstrate hexagonal close packing and cubic close packing. Atoms can be packed together in several different arrangements, and models are used to represent these arrangements. Models that represent how atoms pack together can be similar in type to the space-filling kind. Instead, the colours of these models were chosen for how good they would look in black and white photographs.(1) The set comes with scale cards by Gallenkamp for estimating the size of the molecules (20mm equivalent to 0.1nm). The models themselves are made of a rigid plastic, with a colour scheme that is different from the recommendations set out by the Institute of Physics. The company Griffin and George had this name only between 1954-1957, so these sets of models can be closely dated to this time. in 1952 and underwent several improvements over the next fourteen years. Griffin and George's Courtauld Atomic Models set is extremely well known amongst students of the period. This set of space-filling models in the Whipple Museum's collection was made by Griffin and George, a company that designed mass-produced models for students learning chemistry. Courtauld space-filling models by Griffin and George Space-filling models use a measurement known as the van der Waals radius to give the accurate size of each type of atom, based on the density of electrons around them. Ball and spoke representations are much better for showing this information. However, space-filling models make it difficult to see how the atoms bond together and prevents seeing the structure of the whole molecule clearly. Chemistry students use space-filling models to help when visualising whether the shape of certain bulky structures will prevent them reacting with other molecules.
